Safety and Tolerability, Pharmacokinetics, and Pharmacodynamics of ACT017, an Antiplatelet GPVI (Glycoprotein VI) Fab
Voors-Pette C, Lebozec K, Dogterom P, Jullien L, Billiald P, Ferlan P, Renaud L, Favre-Bulle O, Avenard G, Machacek M, Pletan Y, Jandrot-Perrus M
2019 • Arterioscler Thromb Vasc Biol • [pdf] • [Editorial]
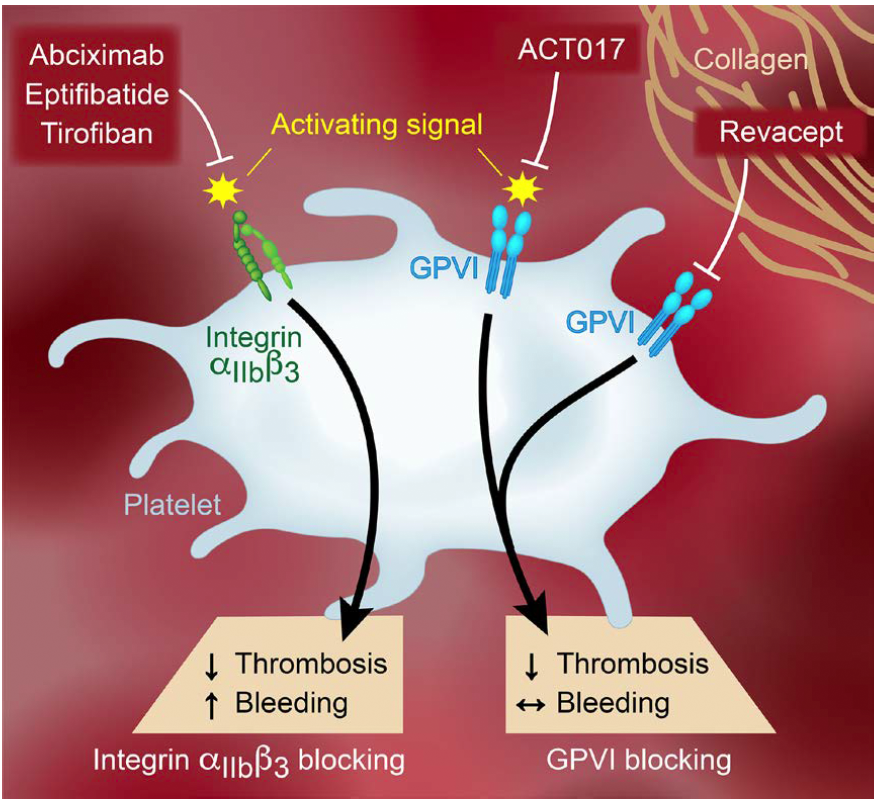
Objective– ACT017 is a novel, first in class, therapeutic antibody to platelet GPVI (glycoprotein VI) with potent and selective antiplatelet effects. This first-in-human, randomized, placebo-controlled phase 1 study was conducted to evaluate the safety, tolerability, pharmacokinetics, and pharmacodynamics of ACT017 in healthy subjects.
Approach and Results– Six cohorts of 8 healthy male and female subjects each received ascending single doses of ACT017 (n=6) or placebo (n=2) as a 6-hour intravenous infusion, with (1/4) of the total dose administered within 15 minutes and the rest of the dose ((3/4) of the total dose) administered within 5 hours and 45 minutes. The 6 investigated doses ranged from 62.5 to 2000 mg. All doses of ACT017 were well tolerated, and no serious adverse events occurred during the study. None of the subjects reported an infusion site reaction. Template bleeding time was not affected in a clinically significant manner by any of the ACT017 doses. Plasma concentrations, determined by liquid chromatography-tandem mass spectrometry, increased linearly with the dose received as were the established pharmacokinetics values. There was no change in the platelet count, platelet GPVI expression assessed by flow cytometry, or plasma levels of soluble GPVI assessed by ELISA. In contrast, administration of ACT017 inhibited collagen-induced platelet aggregation measured by light transmission aggregometry on platelet-rich plasma, and the extent and duration of the effect were dose-dependent.
Conclusions– The novel antiplatelet agent ACT017 has consistent pharmacokinetic/pharmacodynamic properties and favorable safety and tolerability profiles warranting further clinical development.